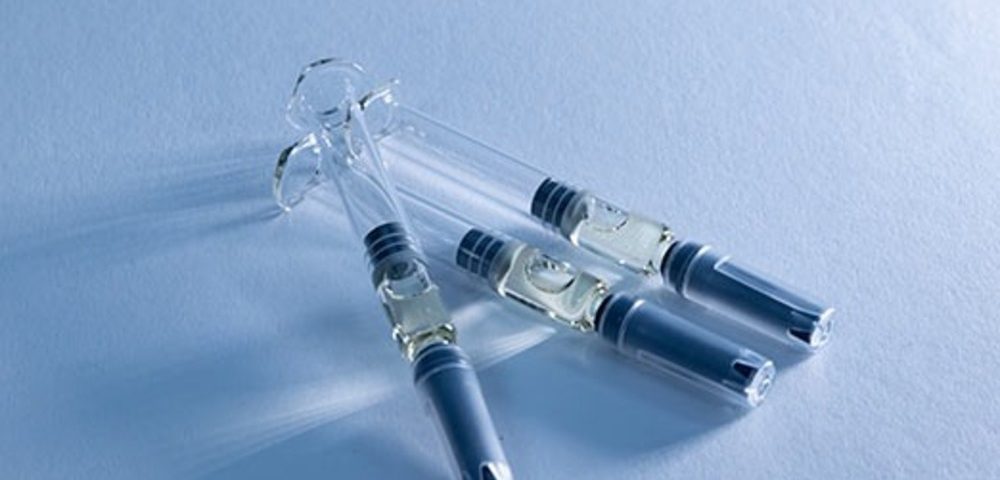
Maisons-Alfort, France
At Adragos Pharma’s Maisons-Alfort facility in France, we manufacture sterile drug products at commercial scale, specialising in prefilled syringes (PFS) and liquid and lyophilised vials. The site combines high-speed aseptic filling, large-scale freeze-drying, advanced visual inspection, dedicated packaging halls and robust logistics. Operating to stringent GMP standards, supported by CIP/SIP and active RABS technologies, our manufacturing site is built to deliver reliable quality, compliance and continuity of supply for global programmes.
Dosage Forms and Manufacturing Services

Prefilled Syringes (PFS) Manufacturing
Maisons-Alfort site supports multiple PFS presentations and high-throughput production:
• 2 aseptic filling lines dedicated to PFS
• PFS types: Standard, Preventis™, Eris™
• Filling speed: up to 540 syringes/min
• Formats: 0.5 mL and 1 mL
• Controlled aseptic environments with active RABS
• Integrated compounding and automated cleaning/sterilisation (CIP/SIP)
• Capacity support up to 150 million units/year (programme dependent)

Liquid & Lyophilised Vials Manufacturing
The site manufactures both liquid and lyophilised vials with industrial freeze-drying capacity:
• 1 aseptic filling line and 2 large freeze dryers
• Annual capacity: up to 6 million vials/year
• Filling: up to 300 vials/min
• Freeze-drying: up to 70,000 vials (format dependent)
• Crimping: up to 350 vials/min
• Formats: 6R, 10R, 7 mL, 10 mL, 15 mL, 22 mL
• Controlled aseptic environments with active RABS
• Integrated compounding and CIP/SIP cleaning/sterilisation

Supporting Services
• Visual inspection: automated and semi-automated systems for syringes and vials
• Packaging and assembly: syringes, vials, safety devices and complex kitting
• Storage and logistics: three-warehouse model with on-site and external capacity
• Quality and testing: GMP controls across aseptic processing, CIP/SIP and RABS environments
Key Capabilities
Maisons-Alfort site is designed to combine speed, scale and quality across a broad sterile portfolio. High-capacity aseptic filling and industrial freeze-drying enable flexibility for both syringes and vials, while integrated inspection systems support consistent product quality at throughput. Dedicated packaging halls handle everything from standard presentations to complex kitting, and a robust logistics model supports reliable global supply.

Visual Inspection for Syringes and Vials
Most visual inspection at the site is automated to secure consistent quality at scale.
Prefilled Syringes Inspection
- • 2 inspection lines
- • 100% automated inspection
- • Maximum capacity: up to 600 syringes/min
Vials Inspection
• Maximum capacity: up to 100 vials/min
• Semi-automated inspection
Packaging and Assembly
A dedicated packaging hall supports both syringes and vials, from standard presentations to complex kit.
Packaging for Prefilled Syringes
• 6 packaging lines dedicated to PFS
5 thermoforming packaging lines for plain and safety syringes
1 plastic-free line
• Speed: up to 360 syringes/min
• Kit formats: 2- to 76-syringe packs, including Blister-to-Box (BIB) options
Packaging for Vials
• 2 packaging lines dedicated to vials
1 plastic-free line
1 blister line
• Cartoning: up to 130 vials/min
• Thermoforming: up to 75 blisters/min for combined kits
Storage and Logistics
Maisons-Alfort site is supported by a scalable, three-warehouse model to ensure continuity of supply and flexibility for scale-up. Warehouse operations are GMP-qualified with temperature mapping and continuous temperature monitoring to support controlled distribution.
Download Our Brochure Now!
We have prepared a brochure to help you discover our facility in Maisons-Alfort.

Key Information
Location: Maisons-Alfort, France
SQM: 30,800 m²
Foundation Year: 2017
FTE (Employees): 452 FTE

Certifications
The site operates under EU GMP with certification by ANSM (France) and maintains strong audit readiness. Maisons-Alfort site supports global regulatory programmes and is positioned as an FDA-ready site.
• France: ANSM
• Asia: NMPA (China), MFDS (South Korea), SFDA (Saudi Arabia), NCE (Kazakhstan), TITCK (Turkey)
• Africa: NAFDAC (Nigeria), NDA-UG (Uganda), PPB (Kenya)
• Others: ANVISA (Brasil), MHRF (Russia), Belarus

Expertise
• Prefilled Syringes (PFS)
• Lyophilised and Liquid vials: Aseptic filling, freeze-drying and sealing
• Visual inspection
• Packaging & assembly
• Storage & logistics
• Quality & testing

Meet our Site Head
Thomas Béchu
Thomas is the Head of Adragos Maisons-Alfort, where he leads sterile manufacturing operations with a focus on Prefilled Syringes (PFS) and liquid and lyophilised vials. With more than 16 years of experience in production and packaging at Sanofi, he has managed large teams and complex aseptic processes. At Maisons-Alfort site, he drives quality, efficiency, and reliable global supply.
For a more comprehensive look into Thomas’ professional profile, connect with him on LinkedIn.
Latest Articles
Read more about what’s happening at Adragos Maisons-Alfort.